字體:小 中 大
字體:小 中 大 |
|
|
|
| 2020/09/11 14:32:53瀏覽986|回應0|推薦0 | |
|
thermodynamic properties of pure gas and ideal gas mixture 1. isothermal G-P relation of pure gas, dG=-SdT+VdP at fixed T ∫dG=∫VdP=∫(RT/P)dP=∫ᴘ₀ᴾ RTdlnP, ∆G=G-G⁰=RT(lnP-lnP₀) i.e. Choose P₀=1 atm, pure gasG⁰=0 dG=RTdlnP → G=G⁰+RTlnP 2. ideal gas mixture, at fixed T, P and V
nₐ+nᵇ+nᶜ+....=nᵀ=∑nᵢ, Xᵢ= nᵢ/nᵀ, i=a,b,c,....molar fraction
partial pressure pₐ, pᵇ, pᶜ.... pᵢ=XᵢP partial molar quantity: Qᵢ≡(∂Q∕∂nᵢ)ᴛ,ᴘ,nʲ... e.g. V, S, U, H, A, G; Gᵢ≡(∂G∕∂nᵢ)ᴛ,ᴘ,nʲ...=μᵢ chemical potential §5-6/5-7 G=G(T,P,nᵢ,nʲ...) dG=-SdT+VdP+(∂G∕∂nᵢ)ᴛ,ᴘ,nʲ...dnᵢ+(∂G∕∂nʲ)ᴛ,ᴘ,nᵢ...dnʲ+....=-SdT+VdP+∑(∂G∕∂nʲ)ᴛ,ᴘ,ᶜₒₘₚdnᵢ =-SdT+VdP+∑ μᵢdnᵢ 限制條件ᴛ,ᴘ,nʲ...G與 Qᵢ相同 dU=TdS-PdV+∑(∂U∕∂nʲ)S,ᴠ,ᶜₒₘₚdnᵢ, (∂U∕∂nʲ)S,ᴠ,ᶜₒₘₚ≠ Uᵢ=(∂U∕∂nᵢ)ᴛ,ᴘ,nʲ... dH=TdS+VdP+∑(∂H∕∂nʲ)S,ᴘ,ᶜₒₘₚdnᵢ, (∂H∕∂nʲ)S,ᴘ,ᶜₒₘₚ≠ Hᵢ=(∂H∕∂nᵢ)ᴛ,ᴘ,nʲ... dA=-SdT-PdV+∑(∂A∕∂nʲ)ᴛ,ᴠ,ᶜₒₘₚdnᵢ, (∂A∕∂nʲ)ᴛ,ᴠ,ᶜₒₘₚ≠ Aᵢ=(∂A∕∂nᵢ)ᴛ,ᴘ,nʲ.. 3. pure gas (∂G∕∂P)ᴛ=V gas mixture: for i component, (∂Gᵢ∕∂P)ᴛ,nᵢ,nʲ...= Vᵢ pf: (∂G∕∂P)ᴛ,ᶜₒₘₚ=V, Vᵢ≡(∂V∕∂nᵢ)ᴛ,ᴘ,nʲ...=(∂[(∂G∕∂P)ᴛ,ᶜₒₘₚ]/∂nᵢ)ᴛ,ᴘ,nʲ...=(∂[(∂G∕∂nᵢ)ᴛ,ᴘ,nʲ...]/∂P)ᴛ,ᶜₒₘₚ =(∂Gᵢ∕∂P)ᴛ,ᶜₒₘₚ... ⸫(∂Gᵢ∕∂P)ᴛ,nᵢ,nʲ...= Vᵢ 因此pure gas的Gibbs free energy式子推導都可適用於gas mixture. Pure gas: dG=RTdlnP → G=G⁰+RTlnP vs. Gas mixture: dGᵢ=RTdlnpᵢ → Gᵢ=Gᵢ⁰+RTlnpᵢ
Vᵢ=(∂Gᵢ∕∂P)ᴛ,ᶜₒₘₚ... → ∫ᴳᵢᵒᴳͥdGᵢ =∫ᴘᵢpᵢVᵢdP=∫ᴘᵢpᵢ(XᵢRT/pᵢ)∙(dpᵢ/Xᵢ)=∫ᴘᵢpᵢ RTdlnpᵢ → Gᵢ-Gᵢ⁰=RT(lnpᵢ-lnPᵢ)
i.e. Vᵢ≡(∂V∕∂nᵢ)ᴛ,ᴘ,nʲ...=[∂(nᵢRT/P)∕∂nᵢ]ᴛ,ᴘ,nʲ...=RT/P=RT/(pᵢ/Xᵢ)=XᵢRT/pᵢ
⸫∆Gᵢ=Gᵢ-Gᵢ⁰=RTlnpᵢ choose Pᵢ=1 atm, ∆Gₘᵢₓ=∑(nᵢ∆Gᵢ+nʲ∆Gʲ+...)
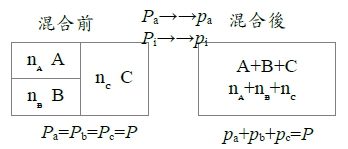
i=a,b,c... 混合前Gₐ=Gₐ⁰+RTlnPₐ → Gᵢ=Gᵢ⁰+RTlnPᵢ(通式)
混合後Gₐ=Gₐ⁰+RTlnpₐ → Gᵢ=Gᵢ⁰+RTlnpᵢ(通式)
∆Gᵢ=Gᵢ-Gᵢ=RTln(pᵢ/Pᵢ)=RTln(pᵢ/P)= RTlnXᵢ
∆Gₘᵢₓ=nₐ∆Gₐ+nᵇ∆Gᵇ+nᶜ∆Gᶜ+...=∑nᵢ∆Gᵢ ⸫∆Gₘᵢₓ=RT∑nᵢlnXᵢ or ∆Gₘᵢₓ=RT∑XᵢlnXᵢ<0 ⸪Xᵢ<1
∆Hₘᵢₓ=∑nᵢ∆Hᵢ=∑nᵢ(Hᵢ-Hᵢ)
Gᵢ已知, 求Hᵢ=? 利用Gibbs-Helmholtz eq. ⸫[∂(Gᵢ/T)∕∂T]ₚ=-Hᵢ/T²
⸪ Gᵢ=Gᵢ⁰+RTlnpᵢ → Gᵢ/T=Gᵢ⁰/T+Rlnpᵢ → [∂(Gᵢ/T)∕∂T]ₚ=[∂(Gᵢ⁰/T)∕∂T]ₚ+[∂(Rlnpᵢ)∕∂T]ₚ i.e.pᵢ=XᵢP
→ -Hᵢ/T²=-Hᵢ⁰/T² → Hᵢ=Hᵢ⁰, 同理 Hᵢ=Hᵢ⁰ ⸫ Hᵢ-Hᵢ=0, ∆Hₘᵢₓ=0
∆Gₘᵢₓ=∆Hₘᵢₓ-T∆Sₘᵢₓ at fixed T, ∆Sₘᵢₓ=-∆Gₘᵢₓ/T=-R∑nᵢlnXᵢ or ∆Sₘᵢₓ=- R∑XᵢlnXᵢ>0
∆Vₘᵢₓ=∑nᵢ∆Vᵢ=∑nᵢ(Vᵢ-Vᵢ)=0
Vᵢ≡(∂V∕∂nᵢ)ᴛ,ᴘ,nʲ...=[∂(nᵢRT/P)∕∂nᵢ]ᴛ,ᴘ,nʲ...=RT/P, Vᵢ=nᵢRT/Pᵢ=nᵢRT/P → Vᵢ=RT/P
∆Uₘᵢₓ=? ⸪∆Hₘᵢₓ=∆Uₘᵢₓ+P∆Vₘᵢₓ ⸫∆Uₘᵢₓ=0
conclusion: one mole ∑Xᵢ=1
∆Gₘᵢₓ=RT∑XᵢlnXᵢ, ∆Sₘᵢₓ=- R∑XᵢlnXᵢ, ∆Hₘᵢₓ=0, ∆Vₘᵢₓ=0, ∆Uₘᵢₓ=0 ideal solution的結果相同
§real gas properties
pure(ideal) gas dG=RTdlnP vs. Real gas dG≡RTdlnf f≠P
gas mixture dGᵢ=RTdlnpᵢ vs. dGᵢ=RTdlnfᵢ (混合前fᵢ⁰; 混合後fᵢ)
Gᵢ=Gᵢ⁰+RTlnfᵢ let fᵢ⁰=1 atm, ∆Gᵢ=Gᵢ-Gᵢ⁰=RTlnfᵢ, ∆Gₘᵢₓ=RT∑Xᵢln(fᵢ/f)
|
|
| ( 知識學習|隨堂筆記 ) |