

 字體:小 中 大
字體:小 中 大 |
|
|
|
| 2016/09/22 00:46:04瀏覽2034|回應0|推薦29 | |
這學期技能課,我又輪到了女性生殖器官內診,以及乳房觸診。 有學生反應~~~可是老師,上學期一位乳外老師說現在不推動乳房處自我觸診了。為此趕快查閱資料,回應學生約略如下,請參考之,但是有問題, 還是請教乳房外科專科醫師 我找到的有關自我觸診篩檢無效文章列於最下; 另附2個自我篩檢影片 1. 目前"美國乳癌協會"並没說要不推廣女性自我檢查 http://www.breastcancer.org/symptoms/testing/types/self_exam 有研究認為導致"驚嚇過度/防禦性醫療.....過多不必要的切片"
改成白話文:研究無法證實有助,但我們不敢說自我檢查真的没意義? (原文:Clinical breast exam and breast self-exam Research does not show a clear benefit of physical breast exams done by either a health professional or by yourself for breast cancer screening. Due to this lack of evidence, regular clinical breast exam and breast self-exam are not recommended. Still, all women should be familiar with how their breasts normally look and feel and report any changes to a health care provider right away.) 3. 目前台灣, 或說全世界, 還有些特別生態: 1) 健檢公司/中心,傾向推銷全身都推銷你檢查,所以20-30歲的妳可能幾乎不可能得乳癌, 還是带着陰影每半年檢查囊腫, 良性腫瘤,害怕之餘也浪费健保資源,負責追蹤的醫師也無法拒绝患者的檢查,因為凡事都有”萬一” 2) 台湾乳癌發生年龄早西方十年,發生率年年攀升。少數乳癌患者甚至年輕到20-30歲,沒錢健檢者,自我檢查或可以讓她們提高生存機率。還有諱疾忌醫者我們也多,自我檢查至少讓40-50以上女性有機會察覺並之道應該就醫 3) 美國癌症協會建議也分成一般人, 與高危險者, 他們高危險者年年乳房MRI.....台灣目前還停留在對東方女性較不敏感的乳房攝影 (http://www.hpa.gov.tw/BHPNet/web/HealthTopic/TopicArticle.aspx?No=201007200001&parentid=201007190003) , 通常健保也提供乳超互補(還好台灣的乳房超音波算普及與技術精進 (異常者則依BIRAD分級追蹤或切片)國民健康署: 篩檢何處去?國民健康署補助45-69歲婦女及40-44歲具乳癌家族史(指祖母、外婆、母親、女兒、姊妹曾有人罹患乳癌)婦女每2年1次乳房X光攝影檢查 (美原文:For women at higher than average risk Women who are at high risk for breast cancer based on certain factors should get an MRI and a mammogram every year. This includes women who: · Have a lifetime risk of breast cancer of about 20% to 25% or greater, according to risk assessment tools that are based mainly on family history (such as the Claus model – see below) · Have a known BRCA1 or BRCA2 gene mutation · Have a first-degree relative (parent, brother, sister, or child) with a BRCA1 or BRCA2 gene mutation, and have not had genetic testing themselves · Had radiation therapy to the chest when they were between the ages of 10 and 30 years · Have Li-Fraumeni syndrome, Cowden syndrome, or Bannayan-Riley-Ruvalcaba syndrome, or have first-degree relatives with one of these syndromes · For women at average risk These guidelines are for women at average risk for breast cancer. Women with a personal history of breast cancer, a family history of breast cancer, a genetic mutation known to increase risk of breast cancer (such as BRCA), and women who had radiation therapy to the chest before the age of 30 are at higher risk for breast cancer, not average-risk. (See below for guidelines for women at higher than average risk.) Women ages 40 to 44 should have the choice to start annual breast cancer screening with mammograms if they wish to do so. The risks of screening as well as the potential benefits should be considered. Women age 45 to 54 should get mammograms every year. 我們還是兩年一次, 除非異常 Women age 55 and older should switch to mammograms every 2 years, or have the choice to continue yearly screening. Screening should continue as long as a woman is in good health and is expected to live 10 more years or longer. All women should be familiar with the known benefits, limitations, and potential harms associated with breast cancer screening. They should also be familiar with how their breasts normally look and feel and report any changes to a health care provider right away. 4. 西方研究(2006, 猶太人; 2003, 加拿大人)不適合東方女性 我們乳腺組織多,若再有纖維化病變,早期發現乳癌比西方女性困難。命是妳我的。根據西方國家的指引,我們都不要平日自摸?敝人認為言之過早,沒有人單獨針對乳腺較多脂肪較少的東方女性作大規模研究。(當然, 通常脂肪多較易觸診出異常) (2012研究也顯示:印第安婦女對自我檢查知識不足…) 5.乳癌分四大類,不同細胞接受器種類狀況都會影響預後。這樣認為不需自我檢查的研究只有助健保支付省錢/花錢,不見得有助降低乳癌死亡率(如乳葉癌Lobular carcinoma佔5-15%)較難能夠以乳房攝影早期發現(越早發現,死亡率越低)。 希望政府多給予一般民眾自我檢查教育, 以上個人淺見,歡迎指正。 乳癌防治基金會有無聲PPT可參考, 或配合Video(英文, 加拿大版)
加拿12 min 較詳細版......即便2003上述他們自己研究也認為"年度醫師乳觸與乳房攝影",罹患後的死亡率自我檢查沒有較低, 它們還是推自我檢查影片....why? 影片還是說, 是要讓你了解乳房一般狀態,無法提早發覺乳癌,理由好奇怪,我想到的是:
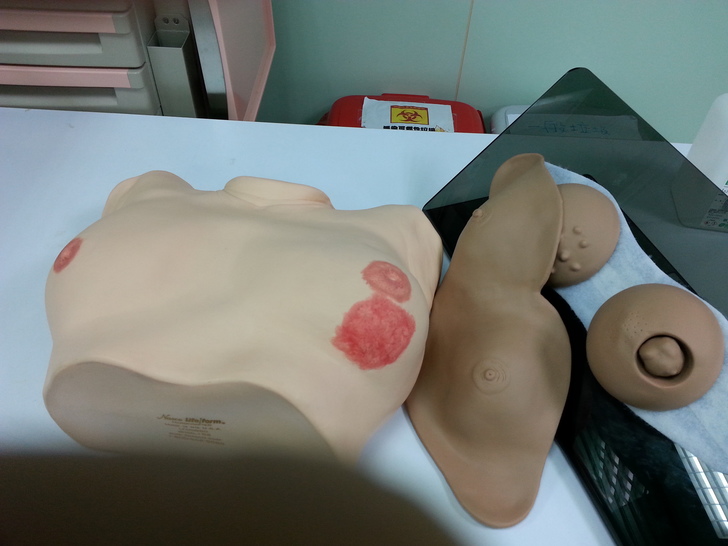
下為練習觸診的教具, 希望能以政府力量推廣給自我檢查者 !!!!!!!!
Psychooncology. 2006 Oct;15(10):873-83. Breast cancer early detection, health beliefs, and cancer worries in randomly selected women with and without a family history of breast cancer.Cohen M1.
AbstractBACKGROUND:Early detection practices (EDP) consist of clinical breast examination (CBE) and mammography. Breast self-examination (BSE) is no longer generally recommended, but many women still perform it. AIMS:To compare EDP, health beliefs, and cancer worries in women with and without a family history of breast cancer in a population-based sample. METHODS:489 women aged 21-60 were randomly sampled from the entire Jewish female population of Israel; 61 (12.5%) had a family history of breast cancer. Participants answered questionnaires by phone, including demographic details, EDP performance, health beliefs, and cancer worries. RESULTS:CONCLUSIONS:(1) A high rate of women did not undergo CBE or mammography screening. Women under 40 with a family history of breast cancer who have never undergone CBE or mammography merit special attention. (2) The change in guidelines on BSE necessitates further study of its over-performance in relation to cancer worries. (3) Interventions are needed to promote attendance for CBE and mammography in younger women with a positive family history. Breast J. 2003 May-Jun;9 Suppl 2:S86-9. Breast cancer mortality in relation to clinical breast examination and breast self-examination.Weiss NS1.
AbstractMammography is the most sensitive available means for early detection of breast cancer, but both clinical breast examination (CBE) and breast self-examination (BSE) have the potential to advance the diagnosis of breast cancer without the expense of a mammography facility. CBE detects about 60% of cancers detected by mammography, as well as some cancers not detected by mammography. There have been no randomized trials comparing breast cancer mortality between women offered and not offered CBE. However, indirect evidence comes from a Canadian study in which women were randomly assigned to CBE alone or CBE plus mammography. Women in the two groups had similar rates of nodal involvement at diagnosis and of breast cancer mortality. Thus if receipt of mammography averts some deaths from breast cancer, the results of this study suggest that CBE has the potential to do so as well. Most studies have found that breast cancers detected by BSE are smaller than those detected without screening and are more likely to be confined to the breast; furthermore, survival after a diagnosis of breast cancer tends to be longer among women who practice BSE than among women who do not. However, neither observational nor randomized studies of BSE provide evidence that this screening modality reduces breast cancer mortality. A recent randomized study in Shanghai, China, found that women assigned to extensive BSE instruction and women assigned to another health intervention had similar distributions of cancer size and stage at diagnosis and similar breast cancer mortality rates. In summary, CBE appears to be a promising means of averting some deaths from breast cancer, whereas BSE appears to have little or no impact on breast cancer mortality. |
|
| ( 知識學習|健康 ) |




 參考資料
參考資料




