字體:小 中 大
字體:小 中 大 |
|
|
||||||||||||
| 2022/02/17 01:34:58瀏覽5573|回應0|推薦1 | ||||||||||||
中重度異位性皮膚炎生物製劑新藥: Tralokinumab
汐止國泰綜合醫院皮膚科主任 美國西北大學芝加哥兒童醫院研究醫師 俞 佑
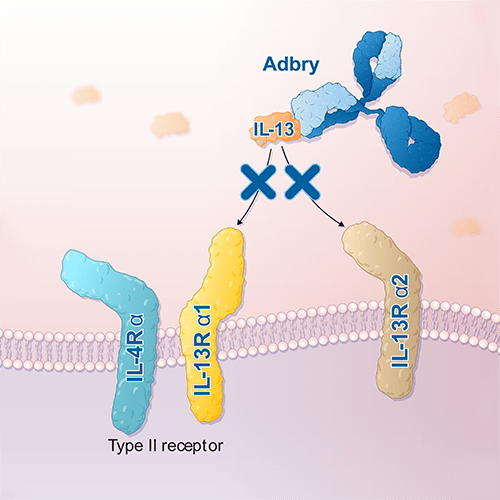
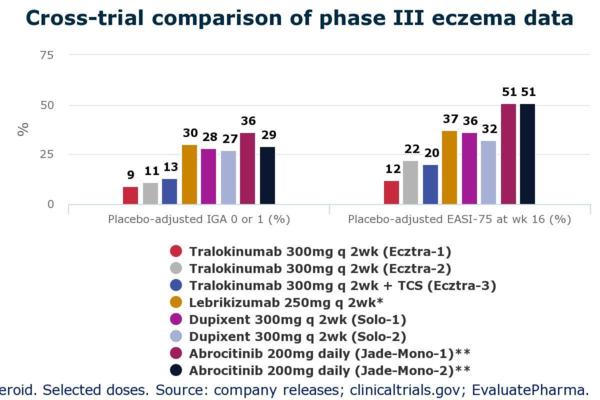
美國FDA已於2021年底正式核准Tralikumab (美國商品名Adbry;美國以外地區商品名Adtralza)作為首個唯一特異性以中重度異位性皮膚炎成人IL-13為標靶的治療藥物。這是針對異位性皮膚炎第二個正式上市的生物製劑(原本只有杜避炎Dupixent (Dupilumab)),因此也獲得醫界高度關注的眼光。
參考資料: 1. https://www.dermatologytimes.com/view/fda-approves-tralokinumab-idrm-for-ad 2. Wollenberg, A et al. “Tralokinumab for moderate-to-severe atopic dermatitis: results from two 52-week, randomized, double-blind, multicentre, placebo-controlled phase III trials (ECZTRA 1 and ECZTRA 2).” The British journal of dermatology vol. 184,3 (2021): 437-449. 3. Silverberg, J I et al. “Tralokinumab plus topical corticosteroids for the treatment of moderate-to-severe atopic dermatitis: results from the double-blind, randomized, multicentre, placebo-controlled phase III ECZTRA 3 trial.” The British journal of dermatology vol. 184,3 (2021): 450-463. 4. https://www.adbry.com/ 5. https://www.linkedin.com/pulse/tralokinumab-how-does-stand-compared-dupilumab-alvaro-moreira 6. https://www.evaluate.com/vantage/articles/news/trial-results/aad-2020-leos-eczema-wildcard-proves-tame
全文完
|
||||||||||||
| ( 知識學習|健康 ) |