字體:小 中 大
字體:小 中 大 |
|
|
|
| 2019/03/29 16:21:30瀏覽113|回應0|推薦0 | |
時間過了約11年,筆者在The Economist這本150年以上歷史的刊物的網上討論區打混好幾年,遇到當時的諾貝爾獎的文章並進行討論,所貼出的autophagy的討論和引用網站登載如下: 原文是在 https://www.economist.com/user/2791934/comments?page=4
Seven tickets to Stockholm Oct 10th 2016, 03:32
This year’s Nobel Science Prizes (NSP) honour researchers devoting themselves to molecular-view progress. Japan’s (J) scientific (SCI) ability catches the world’s breathe for the Yoshinori Ohsumi’s (O) single-handed own.
“Before 2000, only 5 [NSP] were awarded to J researchers, surprisingly few for a rich, high-tech country, while in the last 17 years J is almost keeping up with the USA…” a blogger on the Guardian exclaimed in a comparison to US acme attainment. “Considering that prizes are given years or decades after the discovery, it seems to me [J] is collecting the results of its most [economic affluence’s] era (1960s to late 1980s) and that they havent cut spending for R&D even in the last twenty years of stagnation, recession or weak growth. If so, its a lesson to be learned elsewhere, specially in Europe.”
Since Hideki Shirakawa was rewarded for the discovery (D) of conductive polymers, the 17 Japanese including 2 US citizens shines in the world’s academy during the latest decade. Indeed, in their childhood, either the Showa Financial Crisis or the Recovery to Peace, the cram-based fundamental education was the mainstream in the midst of industrialization. The aggressive college entrance’s exam (CEE) in J, superficially pertinacious subterfuge of drudgery, drives generations to live under didactic social hierarchy. Yet the “Kwantung Army” produces the substantial cornucopia who exemplifies the level-headiness. O san was ranked 1st in the J’s CEE whose seminal observation deserves to be the pièce de résistance for J’s all generation. Besides, J’s academy of college long-time cultivate an indigenous order and formula from the West and the spiritual inheritance of J tradition.
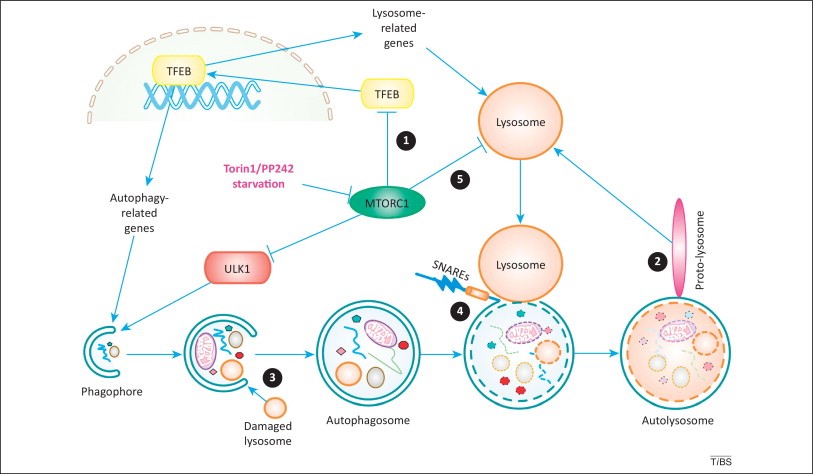
The term “autophagy” (A), derived from the Greek meaning ‘eating of self’, was first coined by Christian de Duve in 1963, largely based on the observed degradation (DE) of mitochondria (MI) and other intra-cellular structures within lysosomes (LY) of rat liver perfused with the pancreatic hormone, glucagon. O started a lab in 1988, focusing on protein (P) DE in the vacuole, an organelle (OR) that corresponds to LY in the human cells (HC). O’s devotion is the protease’s (PR) extensive concept of DE: how HC get rid of larger P complexes and worn-out OR? - since NSP chemistry in 2004.
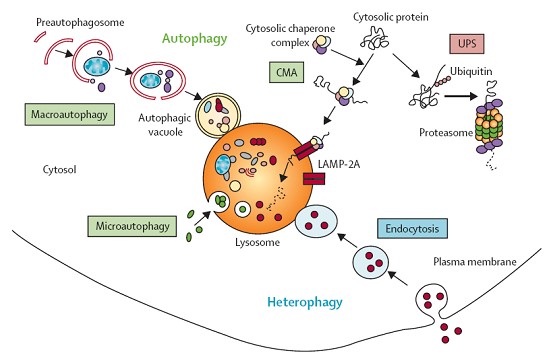
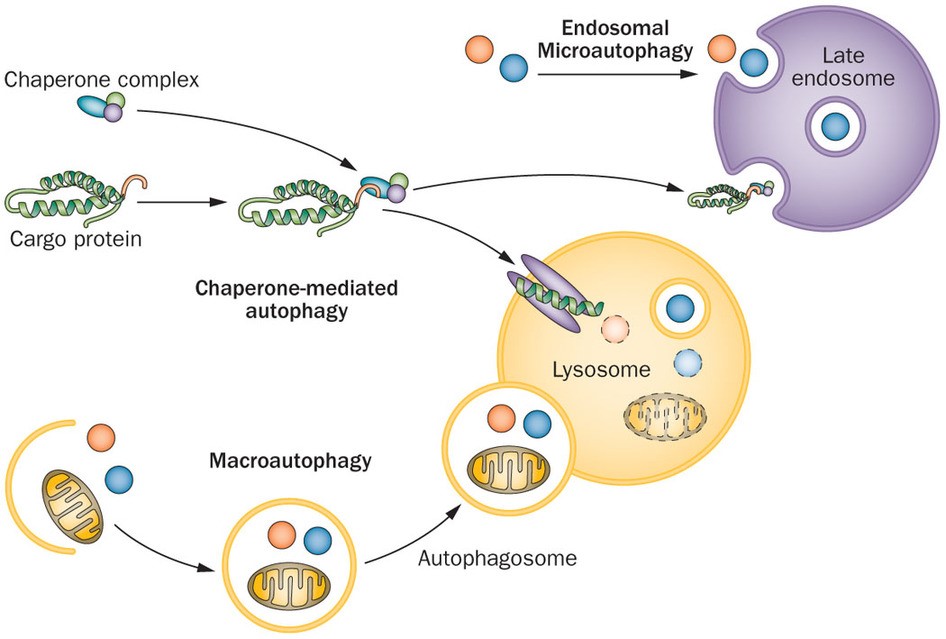
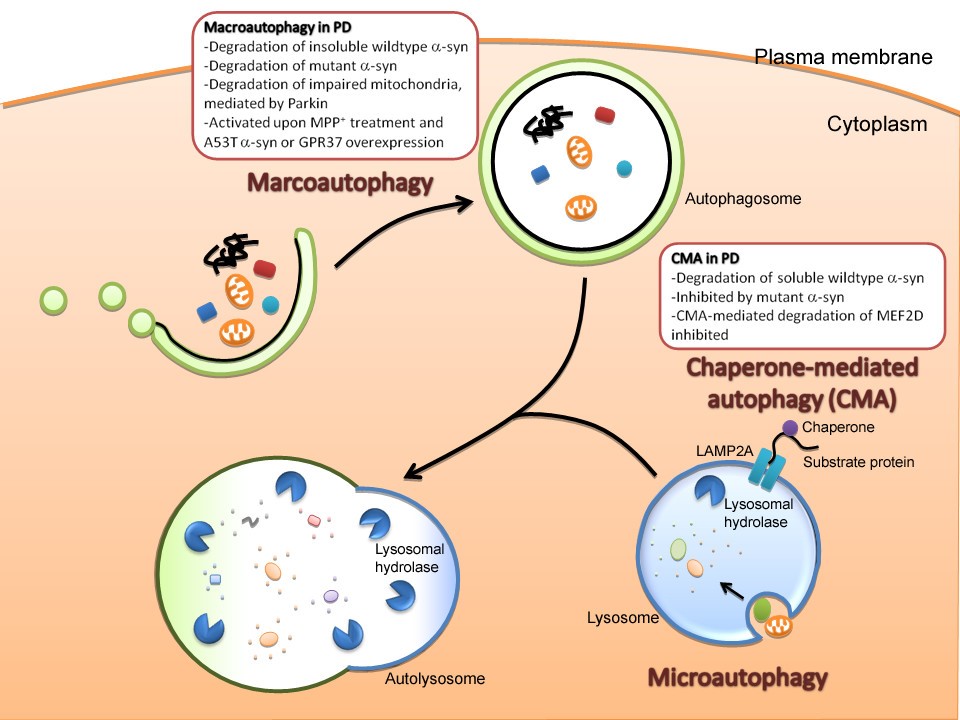
O’s D of A’s relative genes is fulfillment of HC’s life cycle, elucidating another HC death access after apoptosis & necrosis (N). A is divided to 3 types: micro-, Chaperone-mediated & macro-A, all largely related to LY’s participate and LC3B work.
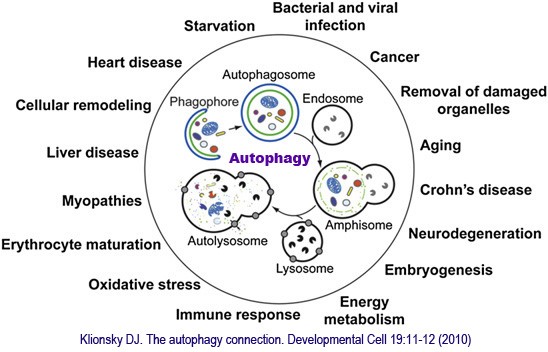
A is the most ancient immune system (IS) that cells deal with regular stress (misfolded PR, aged or defective OR) as well as unexpected disasters (hypoxiconditions, serum starvation, viral infections). The process of A begins with the formation and elongation of isolation membranes, or phagophores. Soon, A-related genes (Atg, 32 types found) excites which helps form the double-membrane autophagosome (DAP). TLR4 induces A in murine macrophage (MA) with LPS’s positive feedback. The cytoplasmic cargo is then sequestered, and DAP fuses with a LY to generate the auto-LY. Finally, DE is achieved through the hydrolases acting within the auto-LY by LY’s acid P. For instance, NLRs in mice, NLDP and RLRs (virus-sensing kinase, VS), Rapamycin (R, bacteria-sensing kinase, BS) in HC or the use of starvation are inducers as well, whilst NLRP4 (VS), 3-MA (BS) are inhibitors. The gamma type (TH1) of Interferon (cytokine, IL) is a potent inducer of A while the IL-4 & 13 (TH2) have been shown to have inhibitory effect. Many of other IL play roles of either promotors or inhibitors with MA, NET and the like working during BS of A.
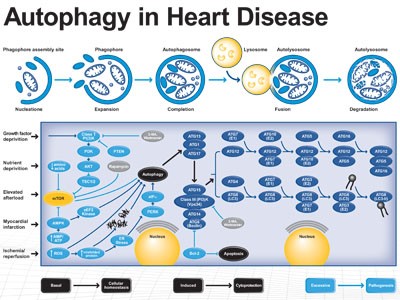
A has become bonafide regulator of both innate and acquired IS. A is implicated in pathogen (PA) sensing, phagocytosis, the removal of intracellular PA and IL production. Also, A promotes cellular senescence and cell surface ‘s antigen presentation (AP), protects against genome instability and prevents N, given a key role in preventing diseases such as cancer, neurodegeneration, cardiomyopathy, diabetes, liver and autoimmune diseases, and infections. By comparison, some human diseases are linked to the dysregulated A process associated to Atg’s deficiency.
In the adaptive IS response, A plays a crucial role in MHC II, MLA’s expressing protein on immune cells’ membrane, during AP whilst MHC I few involved in A except for MA and so on. For instance, R can strengthen MHC II’s AP. A is constitutionally in T cells, the activation of which help A’s development and then maintain ER & MI homeostasis like in lymphocytes. Also the likely access exists in B cell, for instance, the A-major differentiation into plasma cells.
Now the academy is curious about whether A’s appearance is driven by nutrient deprivation-induced inhibition of “target of R” and downstream effects on translation of key cell cycle genes, such as cyclin D1, and how Atg-5 & 7 are regulated in ways of either independent or not
Seven tickets to Stockholm Oct 10th 2016, 03:33
Reference and relative pictures: http://www.slideshare.net/ishfaqmaqbool3/autophagy, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2990190/
Recommended 15 Report Permalink 原來的雜誌上的文章: The 2016 Nobel science prizes Seven tickets to Stockholm This year’s awards go to work on nanotechnology, cellular refuse-recycling mechanisms and the applications of topology Oct 8th 2016 | From the print edition
BIGGER is not always better. Anyone who doubts that has only to look at the explosion of computing power which has marked the past half-century. This was made possible by continual shrinkage of the components computers are made from. That success has, in turn, inspired a search for other areas where shrinkage might also yield dividends. One such, which has been poised delicately between hype and hope since the 1990s, is nanotechnology. What people mean by this term has varied over the years—to the extent that cynics might be forgiven for wondering if it is more than just a fancy rebranding of the word “chemistry”—but nanotechnology did originally have a fairly clear definition. It was the idea that machines with moving parts could be made on a molecular scale. And in recognition of this goal Sweden’s Royal Academy of Science this week decided to award this year’s Nobel prize for chemistry to three researchers, Jean-Pierre Sauvage, Sir Fraser Stoddart and Bernard Feringa, who have never lost sight of nanotechnology’s original objective. In this section
Related topics Dr Sauvage’s contribution, for which he won a third of the SKr8m ($930,000) prize money, was to link atoms together in a new and potentially useful way. Conventional molecules are held together by bonds in which electrons from neighbouring atoms pair up. Sometimes (as in the case of benzene) the result is an atomic ring. Dr Sauvage realised that rings of this sort might then be joined with each other in the way that the links of a metal chain are, to create a “supermolecule” that is held together mechanically rather than by conventional chemical bonds. In 1983 his research group at Strasbourg University, in France, managed to make a supermolecule of this sort and, 11 years later, they demonstrated an arrangement, consisting of two such links, that had special properties. Applying energy to it caused one of the links to rotate around the other, creating a species of molecular motor. Small is beautiful Sir Fraser won his third of the prize for work on a similar miniature machine. In 1991 he and his colleagues at Northwestern University in Illinois managed to thread a tiny molecular axle through a ring-shaped molecule. Heating the result caused the ring to slide between the ends of the axle. That produced a molecular shuttle. Since then his group has diversified into other machines, including an atomic-scale lift, artificial muscles and even a simple mechanical computer made of molecule-sized components. The most desired goal of nanotechnology research, however, has always been a motor that rotates around an axle, rather than just sliding up and down it. And it was for creating such a device, in 1999, that Dr Feringa will receive his share of the prize. His insight was to work out how to make the ring spin reliably in a single direction—because a motor that might, at random, turn either way when you start it up is not much use. By 2011 his team at Groningen University, in the Netherlands, had grown sufficiently dexterous to make a “nanocar”. This consists of a molecular chassis connected to four wheels which move the car (very slowly) across a surface. How long it will take to turn any of these inventions into products remains to be seen. Optimists talk of manufacturing molecule-sized machines ranging from drug-delivery devices to miniature computers. Pessimists recall that nanotechnology is a field that has been puffed up repeatedly by both researchers and investors, only to deflate in the face of practical difficulties. There is, though, reason to hope it will work in the end. This is because, as is often the case with human inventions, Mother Nature has got there first. One way to think of living cells is as assemblies of nanotechnological machines. For example, the enzyme that produces adenosine triphosphate (ATP)—a molecule used in almost all living cells to fuel biochemical reactions—includes a spinning molecular machine rather like Dr Feringa’s invention. This works well. The ATP generators in a human body turn out so much of the stuff that over the course of a day they create almost a body-weight’s-worth of it. Do something equivalent commercially, and the hype around nanotechnology might prove itself justified. How cells eat themselves Another example of natural nanotechnology is “autophagy” (from the Greek for “self-eating”). This is the system which breaks up and recycles worn-out cellular components. And the Nobel prize for physiology or medicine, awarded by the Karolinska Institute, went to one of the researchers most involved in discovering how autophagy works: Yoshinori Ohsumi of the Tokyo Institute of Technology. When Dr Ohsumi began his studies biologists did know that autophagy was a two-step process. First, the cellular components to be recycled are enclosed in a fatty membrane, to create a bubblelike vesicle called an autophagosome. Then the autophagosome merges with a second vesicle known as a lysosome. This is filled with digestive enzymes that break up the autophagosome’s contents. They did not, though, know the details. In particular, how autophagosomes formed was a mystery. It is for supplying those details that Dr Ohsumi has been awarded his prize. He began working on the problem in 1988, looking at autophagy in yeast. This is a well-studied organism, often used by biologists to examine fundamental cellular processes. Unfortunately, yeast cells are small. This meant that, to start with, Dr Ohsumi could not easily distinguish the autophagosomes within them under a microscope. He dealt with this difficulty by disabling the manufacture of the lysosomic digestive enzymes. That meant autophagosomes were no longer destroyed, and accumulated to the point where they could be seen and studied. This work, published in 1992, was the key to the rest—the identification of the genes involved in autophagosome assembly, which in turn led to an understanding of how those vesicles come into being. Here, Dr Ohsumi’s choice of yeast paid off. The yeast genome was already well studied when he started his work, and then became one of the first to be elucidated in its entirety as part of the Human Genome Project. By knocking out genes so that they stopped working, and then examining the consequences of their absence, he was able to build up a complete picture of the process by which autophagosomes are assembled. And, though yeast and humans are not closely related, autophagy is such a fundamental cellular process that its course in the two species is more or less the same, so yeast autophagy is a good model of its human equivalent. That is important, because Dr Ohsumi’s work has wider ramifications than merely illuminating an important piece of cellular housekeeping. It also helps explain how invading pathogens bacteria and viruses are dealt with (such unwelcome guests are gobbled up by autophagosomes), and shines light on diseases, including Parkinson’s and some sorts of diabetes, caused when autophagy goes wrong and cellular rubbish accumulates. Understanding autophagy, then, has important practical consequences. The opposite seems true of the subject of the physics prize, as the panel which announced the winners were the first to admit. The panellists emphasised the beauty of the mathematics underlying the prize-winning work and de-emphasised the practical applications—of which, at the moment, there are none. The maths in question is topology, a branch of geometry which deals in “invariants”, such as holes, that can exist in geometric shapes only in discrete, integer numbers (nothing can have half a hole in it). The prizewinners—David Thouless of the University of Washington, in Seattle, Duncan Haldane of Princeton University, in New Jersey, and Michael Kosterlitz of Brown University, in Providence, Rhode Island—have applied topology to materials science and come up with theoretical explanations about the behaviour of unusual states of matter as a result. The all-consuming vortex Intriguingly, all three prizewinners are products of the 20th-century “brain drain” that saw British-born researchers head west to the larger salaries and better laboratories of America. Dr Thouless, who takes home half the prize money, collaborated with Dr Kosterlitz, who shares the other half with Dr Haldane, in the 1970s, when both were still in the old country. The fruit of their collaboration was to overthrow the idea that superconductivity (a phenomenon in which the resistance of an electrical conductor vanishes, usually when it has been cooled to a temperature near absolute zero) could not occur in thin layers of material. It could, according to their calculations, because of the effects of paired vortices within such a layer. Vortices, a type of hole, are topological invariants. It is the liberation of these vortices to move around as a material warms up which destroys superconductivity. Such liberation is, in effect, a phase change from one state of matter to another, just as the liberation by heat of atoms from a crystal lattice causes a phase change from solid to liquid. Dr Thouless then went on, after he had moved to America in the early 1980s, to show that stepwise transitions to and from full superconductivity in the presence of a magnetic field (a phenomenon known as the quantum Hall effect) are also a type of topological invariant. And in the late 1980s, after his own transatlantic migration, Dr Haldane showed that magnetic fields need not be involved in the process at all. Choosing to honour such esoteric stuff this year, in particular, was a surprise to many observers, who had thought the discovery in 2015 of gravitational waves, by an experiment called LIGO, might win. That would have been in the spirit of Alfred Nobel’s will, which refers (see article) to his posthumous awards as being for work from the previous year. For whatever reason, however, the great and good of the Royal Academy of Science, who choose the winners of the physics prize as well as that for chemistry, decided to keep ignoring this part of the prizes’ founding document. From the print edition: Science and technology
筆者2016年聊起autophagy自噬作用時,所引用補充的國內科普文章一篇,其他的筆者在剛剛市面有俄羅斯和美國學者的研究連結 細胞大掃除:自噬作用 Posted on 2009/05/08 in 分子與細胞, 生命科學, 細胞生理 26,903 views Print Friendly
細胞大掃除:自噬作用 (Autophagy) 臺北市立建國高級中學生物科黃慧茹實習老師/國立臺灣師範大學生命科學系張永達副教授責任編輯
細胞本身就可以比喻成小型工廠,許多不同的部門(胞器)在分工合作以維持工廠(細胞)的最佳運作,那如果生產線中出現了不良產品時該怎麼辦呢?沒錯!要有回收或是銷毀系統,所以細胞內也是可藉由自身內部微小的「吸塵器」,清理老舊的蛋白質、失常的胞器以及入侵的微生物,這個過程稱為「自噬作用,autophagy」。
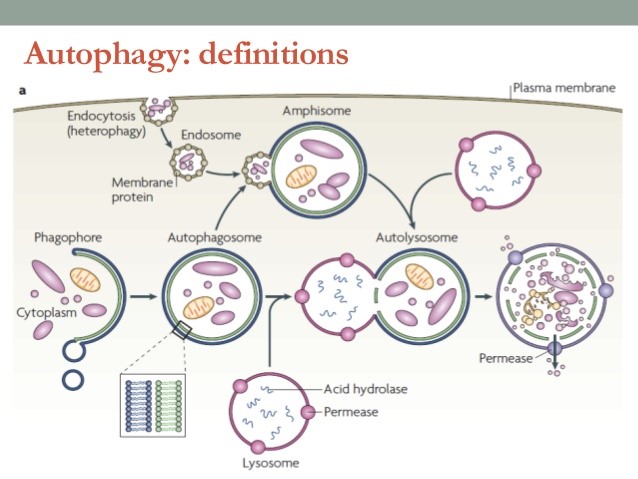
當細胞接受到養分不足、氧氣不足等訊息時,細胞膜上具有訊號接收站能夠將訊息往細胞內回報,此時細胞內的多種蛋白質和脂質先形成彎月狀的雙層膜構造,稱為吞噬泡(phagophore),吞噬泡會藉由增加新的膜逐漸增大,並且將受損的胞器或蛋白質包圍,最後凹陷端關閉成為囊狀構造的自噬體(auto- phagosome),這個自噬體就類似於細胞內的垃圾車,準備將垃圾載往垃圾處理場,垃圾處理場就是溶體,自噬體的外層膜會與溶體(lysosome)的膜融合形成「自噬溶小體,autolysosome」,藉由溶體內水解酵素分解老舊胞器或蛋白質成小分子物質,其中小分子物質如胺基酸則可以再回收利用成為材料,如此完成自噬作用。
不過有時候在嚴重受損的細胞中,垃圾多到垃圾處理場無法負荷時,細胞為顧全大局,原本預備被吞入的受損胞器也可能同時釋放訊號引發細胞凋亡(細胞自殺),細胞凋亡本身也是一連串複雜的訊息傳遞過程,最後會啟動胞內多種水解酵素分解所有胞器及蛋白質,進而造成細胞萎縮死亡。細胞凋亡與自噬作用不同,細胞凋亡就是細胞注定要死亡,但是自噬作用可依細胞內垃圾堆積的程度選擇細胞的存亡與否,所以細胞凋亡與細胞自噬彼此密切又謹慎地相互平衡。
為什麼細胞會有這麼巧妙的安排呢?關於自噬作用的演化成因,科學家推測可能是要應付細胞的飢餓狀態,或做為原始的免疫防禦機制,也可能兩者兼具。所以當我們飢餓卻無法馬上攝食時,身體的各項功能並不會馬上停止,首先是脂肪細胞會先開始將儲備的養分加以分解,最後可能連肌肉細胞的成分都有可能被利用,這些都是要維持必要的代謝作用。
雖然正常細胞會利用自噬作用進行大掃除,許多神經退化性疾病如阿滋海默症的成因就是自噬作用失調,導致細胞內堆積太多垃圾,損壞細胞正常功能。但是,相對的,癌細胞也會利用自噬作用來充飢以延長細胞壽命。因此,有科學家進行抑制腫瘤細胞的自噬作用,作為對抗癌症的策略,不過遺憾的是,抑制腫瘤細胞自噬作用可能會增加癌細胞的基因突變,反而使病人病情惡化,造成對抗癌症更為棘手。
簡而言之,細胞的存亡是取決於自噬作用和細胞凋亡的平衡,其中牽涉許多訊息傳導路徑,目前許多科學家正努力釐清其中調控的機制。
參考資料 1.科學人。第81期。2008年11月號 p.114-122 2.細胞自噬蛋白Atg8表面的功能區域之定性分析論文摘要 何恭憲 2007.11.27臺灣大學碩博士論文網 http://www.cetd.com.tw 3.The WORLD of the CELL 5th edition Becker,Kleinsmith,Hardin 2002 Benjamin Cummings p.353-357 4.新浪新聞網 http://financenews.sina.com/sinacn/304- … 007-07-15/ 1817500029.html 5.百度百科 http://baike.baidu.com/view/294022.html?fromtaglist 6.生命經緯 http://news.biox.cn/content/200701/2007 … 38_27.html 筆者對照後拼的圖 基本模式(兩張)
Nature Liver Muscle
Heart Disease
LAMPA-2A loader
|
|
| ( 心情隨筆|心情日記 ) |